CUNY IRB Review
- Complete CITI Human Subjects Training (yourself and all staff)
- Submit a completed application via IRBManager
Refer to the IRBManager PI manual
IRB Review Categories
- Human Subjects Research Exempt from IRB Review
- Expedited Review of Human Subjects Research
- Convened IRB Review
Please refer to the CUNY HRPP Policies and Procedures page for further details.
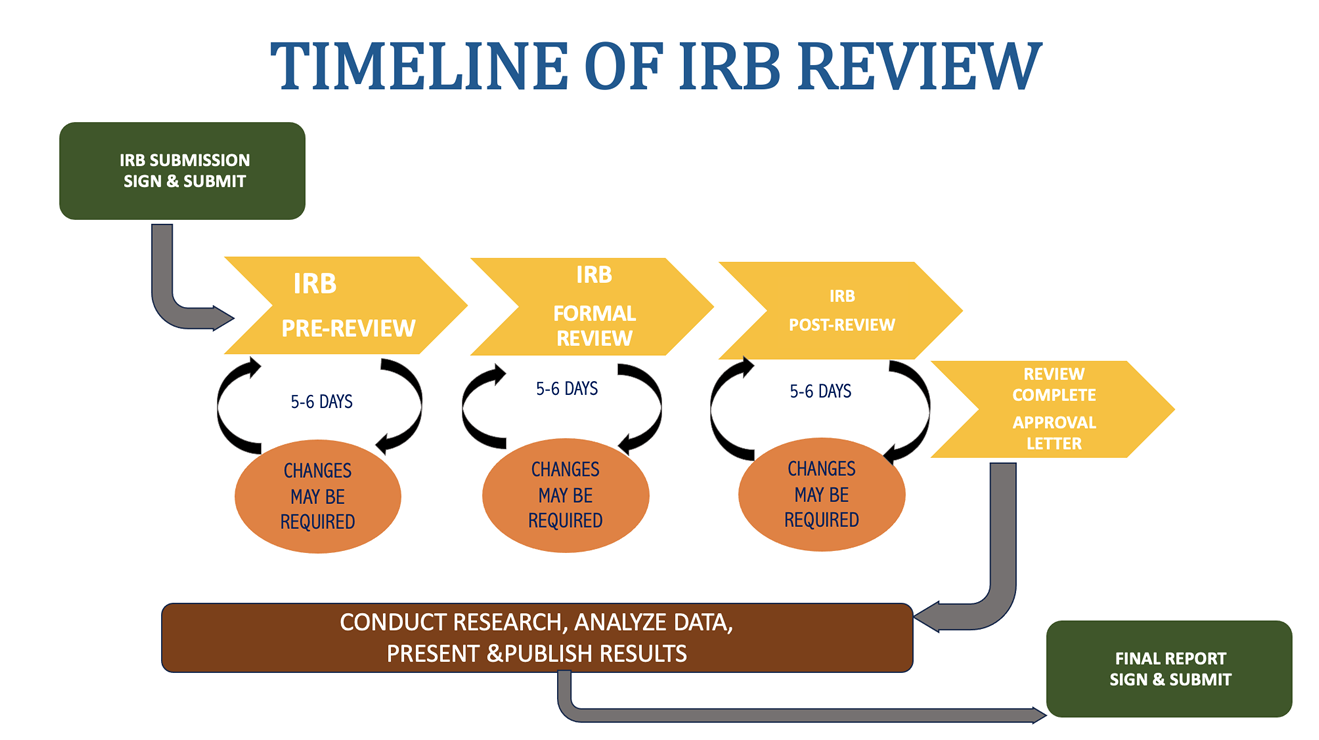
Timeline of IRB Review